Home-Research Shows Sulfur Cathodes Show High Theoretical Promise, but Practical Battery Performance Remains a Major Barrier
Sulfur cathodes remain a prominent area of battery research because they combine low-cost, abundant materials with very high theoretical energy density. A study published inNatureby researchers fromCY University, theCatalonia Institute for Energy Research(IREC), theCatalan Institute of Nanoscience and Nanotechnology(ICN2),ICREA, andFuzhou Universityexamines sulfur-based battery systems through the lens of end-user requirements rather than idealized laboratory performance. Unlike reviews centered mainly on materials advances under controlled conditions, this paper focuses on practical constraints tied to manufacturing and operation, arguing that sulfur-based batteries could support electric mobility, renewable energy integration, and grid stabilization only if performance can be sustained under realistic cell designs.
Lithium-ion batteries still dominate the energy storage market, with average cell costs around €110/kWh, according to the review. That market position remains tied to materials such as nickel, cobalt, and lithium, whose pricing and supply have shown significant volatility. Sulfur offers a route to battery chemistries based on abundant, low-cost, and more widely available materials. Sulfur-based conversion cathodes also have a theoretical specific capacity of 1675 mAh/g and gravimetric energy density approaching 2600 Wh/kg, well above values associated with widely used lithium-ion cathodes including NMC, NCA, LMO, LCO, and LFP. The review, however, makes a clear distinction between theoretical advantage and practical device performance. Many published studies rely on low sulfur loadings of about 1 mg/cm², electrolyte-to-sulfur ratios above 20 µL/mg, and lithium foils thicker than 100 µm. Those conditions are useful for fundamental analysis but tend to overestimate key metrics such as cycle life and capacity retention.
Once sulfur-based cells are evaluated under more commercially relevant conditions, performance drops sharply. At sulfur loadings around 4 to 6 mg/cm², achievable capacity typically falls to about 400 to 600 mAh/g, while cycle life often reaches only 200 to 500 cycles. The review argues that future work should use clearer practical benchmarks, including sulfur loadings above 5 mg/cm², areal capacities above 5 mAh/cm², lean electrolyte conditions below 5 µL/mg, and limited lithium excess. It also notes that many performance targets for electric vehicles and stationary storage may be reachable individually, but not simultaneously within a single practical cell. That distinction is central to the paper’s argument: progress in sulfur batteries has often been measured under conditions that do not reflect commercial requirements.
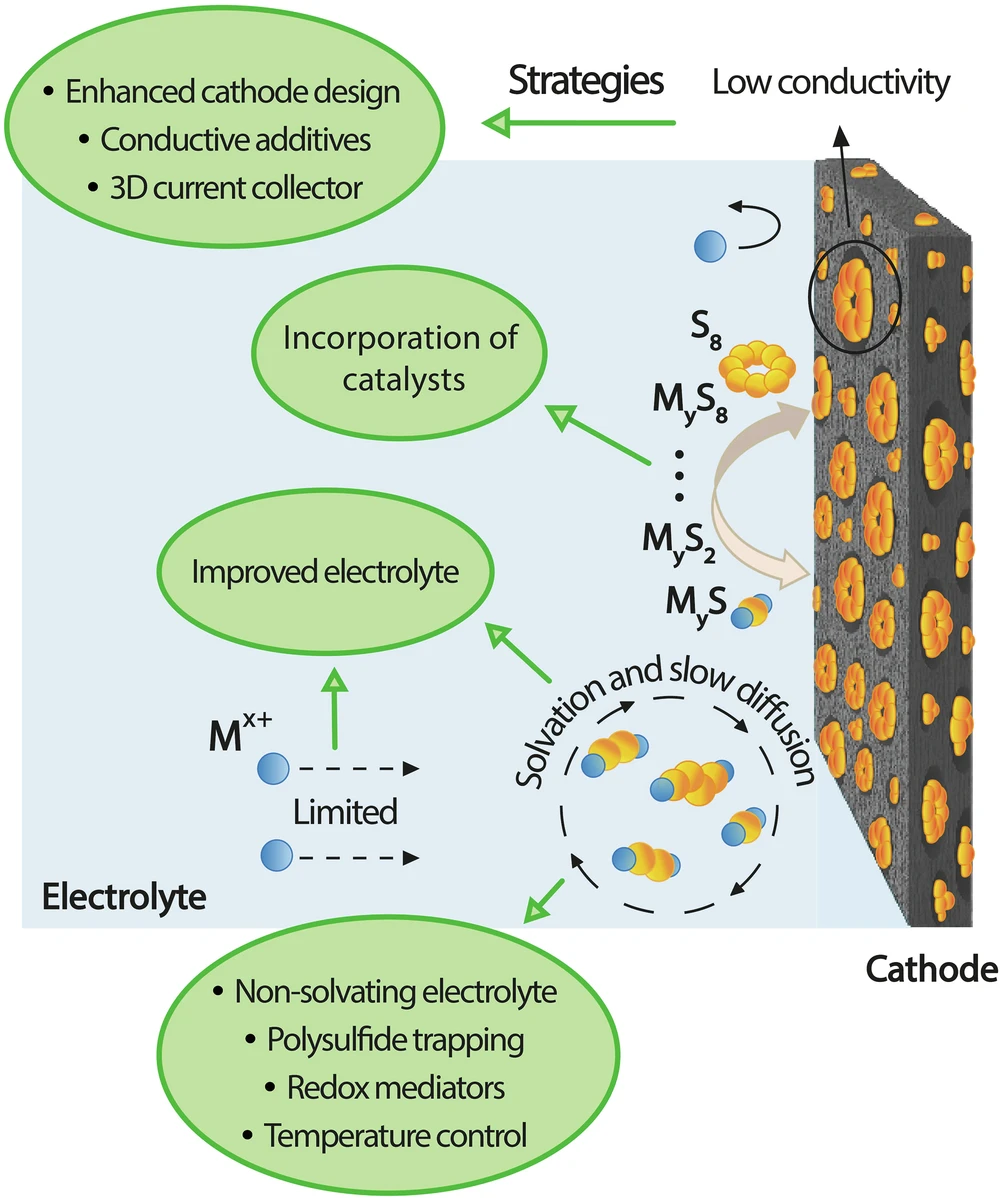
Instead of organizing the field around familiar research themes such as shuttle effects or sulfur reaction kinetics, the review is structured around performance metrics that matter to users. Those include charge and discharge rate, round-trip efficiency, practical energy density, cycling stability, operating temperature range, self-discharge, thermal dissipation, safety, manufacturing cost, infrastructure compatibility, and recycling. Across those categories, the paper describes a set of interlocking constraints. Sulfur and its discharge products are electrically insulating, limiting electron transport. Intermediate polysulfides dissolve into the electrolyte and migrate between electrodes, driving self-discharge, active material loss, and anode degradation. Large volume changes during cycling damage the electrode structure. Ether-based electrolytes such as DOL and DME remain volatile and flammable, while lithium metal anodes introduce dendrite formation and add safety risk. Efforts to raise practical energy density through thicker electrodes, leaner electrolyte use, and lower negative-to-positive capacity ratios intensify those same transport and stability problems.
The review also identifies an expanding commercialization landscape.Lyten, a US battery developer,Gelion, an Anglo-Australian battery company,LG Energy Solution, a South Korean battery manufacturer; Zeta Energy, a US battery materials company,Umicore, a Belgian battery materials supplier, and other firms are advancing sulfur-based systems across sectors that include drones, aerospace, electric vehicles, and stationary storage. At the same time, the paper does not present sulfur batteries as ready to replace lithium-ion cells at scale. Its conclusion is narrower and more specific: sulfur cathodes remain technically compelling, but meaningful progress now depends on testing full cells under practical conditions, reporting energy density at the device level rather than on sulfur mass alone, and developing manufacturing approaches that can support scalable, commercially relevant battery architectures.
The study, titled “Sulfur cathodes for next-generation batteries,” was authored by Alessandra Manzini, Irina Martynova, Jing Yu, Xiaoyu Bi, Jordi Jacas Biendicho, Qing Sun, Jordi Arbiol, Andreu Cabot, and Chaoqi Zhang.
3D Printing Industry is inviting speakers for its 2026 Additive Manufacturing Applications (AMA) series, covering Energy, Healthcare, Automotive and Mobility, Aerospace, Space and Defense, and Software. Each online event focuses on real production deployments, qualification, and supply chain integration. Practitioners interested in contributing cancomplete the call for speakers form here.
Explore the fullFuture of 3D PrintingandExecutive Surveyseries from 3D Printing Industry, featuring perspectives from CEOs, engineers, and industry leaders on theindustrialization of additive manufacturing,3D printing industry trends 2026, qualification, supply chains, andadditive manufacturing industry analysis.
Featured image shows Schematic illustration of the key mechanisms limiting the rate performance of SBBs and the strategies proposed to address them. Image via Nature.
Source: 3D Printing Industry