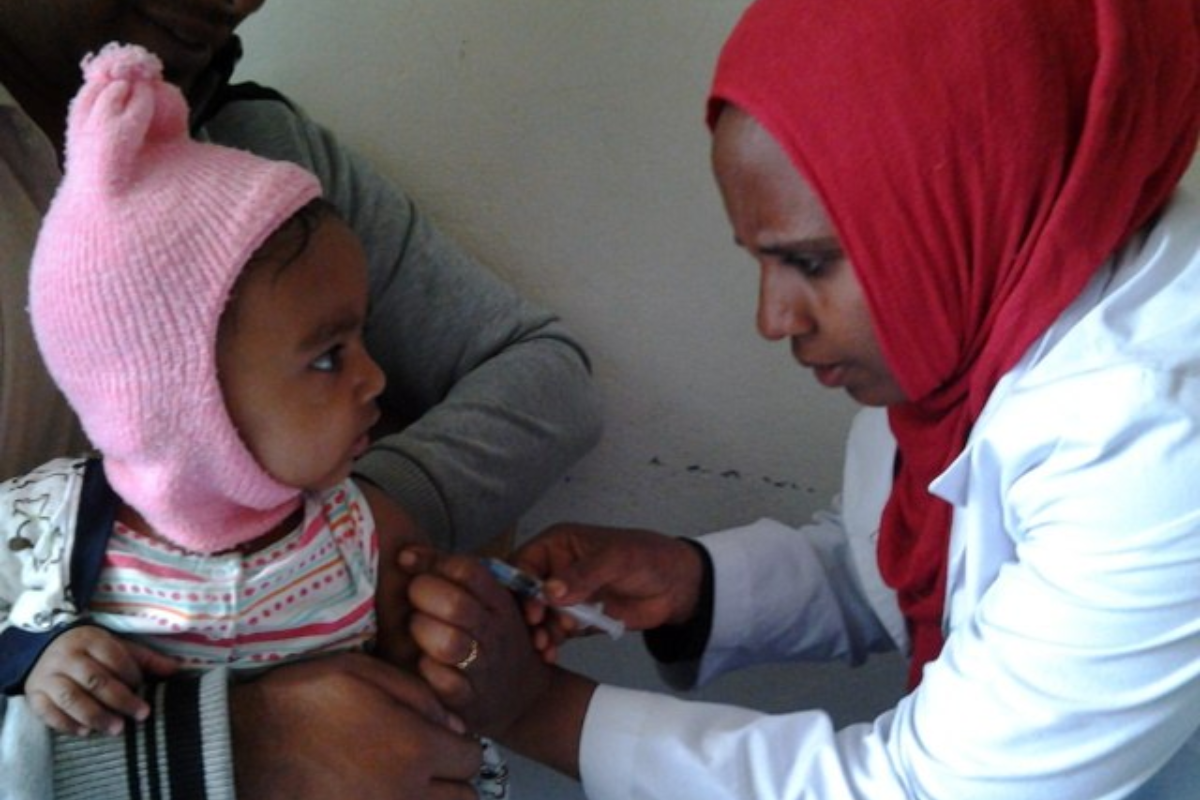
A massive uproar has forced Guinea-Bissau to pull the plug on a US-funded vaccine trial that would have tested 14,000 newborn babies. The suspension came after furious pushback from local officials and international health experts who slammed the research as deeply unethical.
Health MinisterQuinhin Nantotebroke the news during a press conference with the Africa Centres for Disease Control and Prevention late last month. He said the country's ethics committee simply didn't have the 'required technical resources' to properly review such a major study. The announcement followed weeks of mounting anger from critics who accused researchers of exploiting one of the world's poorest countries for a trial that would never fly in wealthier nations.
Dr Magda Robalo didn't mince words when she spoke out against the trial. The former health minister, who spent over 30 years working in global health, including senior roles at the WHO, told the science journal Nature she was appalled her country would even consider such research. 'It's not acceptable and it should not go on,' she said. 'Guinea-Bissauans are not guinea pigs.'
Robalo explained that researchers exploited Guinea-Bissau's weak research infrastructure. She said a small ethics committee within the health ministry knew about the study but failed to inform the national public health institute—the body that should approve such significant experiments.
'We are not second-level citizens,'Robalo said. 'We are not a population to be used for anything that you cannot do in the Global North. We demand respect, despite the fact that we don't have the capacity that we need.'
Planned US-funded baby vaccine trial in Guinea-Bissau blasted by WHOhttps://t.co/3oy6MSlIIN
The controversial study came with a$1.6 million(£1.2 million) price tag from the US Centers for Disease Control and Prevention. Danish researchers at the University of Southern Denmark's Bandim Health Project designed the trial to split babies into two groups randomly. One group would get the hepatitis B vaccine at birth, whilst the other would wait until six weeks old.
Researchers called this a 'unique window of opportunity' since Guinea-Bissau plans to start vaccinating all newborns at birth by 2028. But that's exactly what enraged critics. Why test something we already know works by denying it to thousands of vulnerable babies?
The numbers tell a stark story. More than 12 per cent of adults in Guinea-Bissau carry chronic hepatitis B, according to theWorld Health Organization. When babies catch hepatitis B at birth, roughly 90 per cent develop lifelong infections that can cause liver damage, cirrhosis, and cancer.
The WHO didn't hold back in its criticism. Theorganisation saidit had 'significant concerns regarding the study's scientific justification, ethical safeguards, and overall alignment with established principles for research involving human participants'.
Source: International Business Times UK